 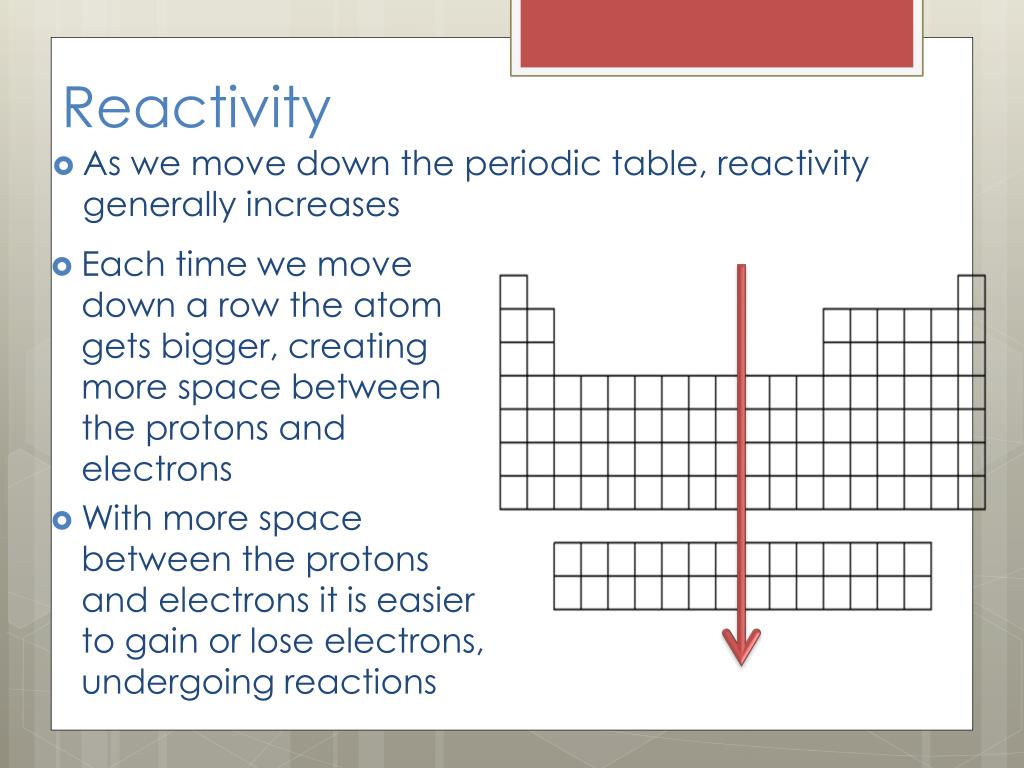
The video below provides an overview of the halogens. Halogen – One of the 5 non-metals in group 7 of the periodic table.ĭisplacement reaction – A reaction in which a more reactive element takes the place of a less reactive element in a compound. Halogens become less reactive as you go down group 7 in the periodic table, because the outer electron shell gets further away from the attraction of the nucleus, and so an electron is gained less easily.Ī less reactive halogen will be displaced by a more reactive one from an aqueous solution of its metal halide.Ĭhlorine + potassium bromide → potassium chloride + bromine Iodine (State of matter at room temperature) Solid (Colour) Grey/ black They form hydrogen halides, which dissolve in water, forming acidic solutions.įluorine (State of matter at room temperature) Gas (Colour) YellowĬhlorine (State of matter at room temperature) Gas (Colour) Greenīromine (State of matter at room temperature) Liquid (Colour) Red/ orange They form molecular compounds with non-metals. Learn about the groupings of metals and nonmetals on the periodic table, features in metallic bonding and reactivity, and their formation of ionic compounds. Skip navigation 0:00 / 3:46 Which Is The Most Reactive Element In The Periodic Table ScienceABC II 2.88K subscribers Subscribe 902 views 3 months ago reactivity periodictable cesium The. They react with metals to form ionic compounds where the halide ion has a charge of -1. They consist of molecules made up of two atoms (diatomic molecules). The halogens have the following properties: 
Group 7 of the periodic table is home to the Halogens. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
